Ch2 atomic orbitals6/21/2023 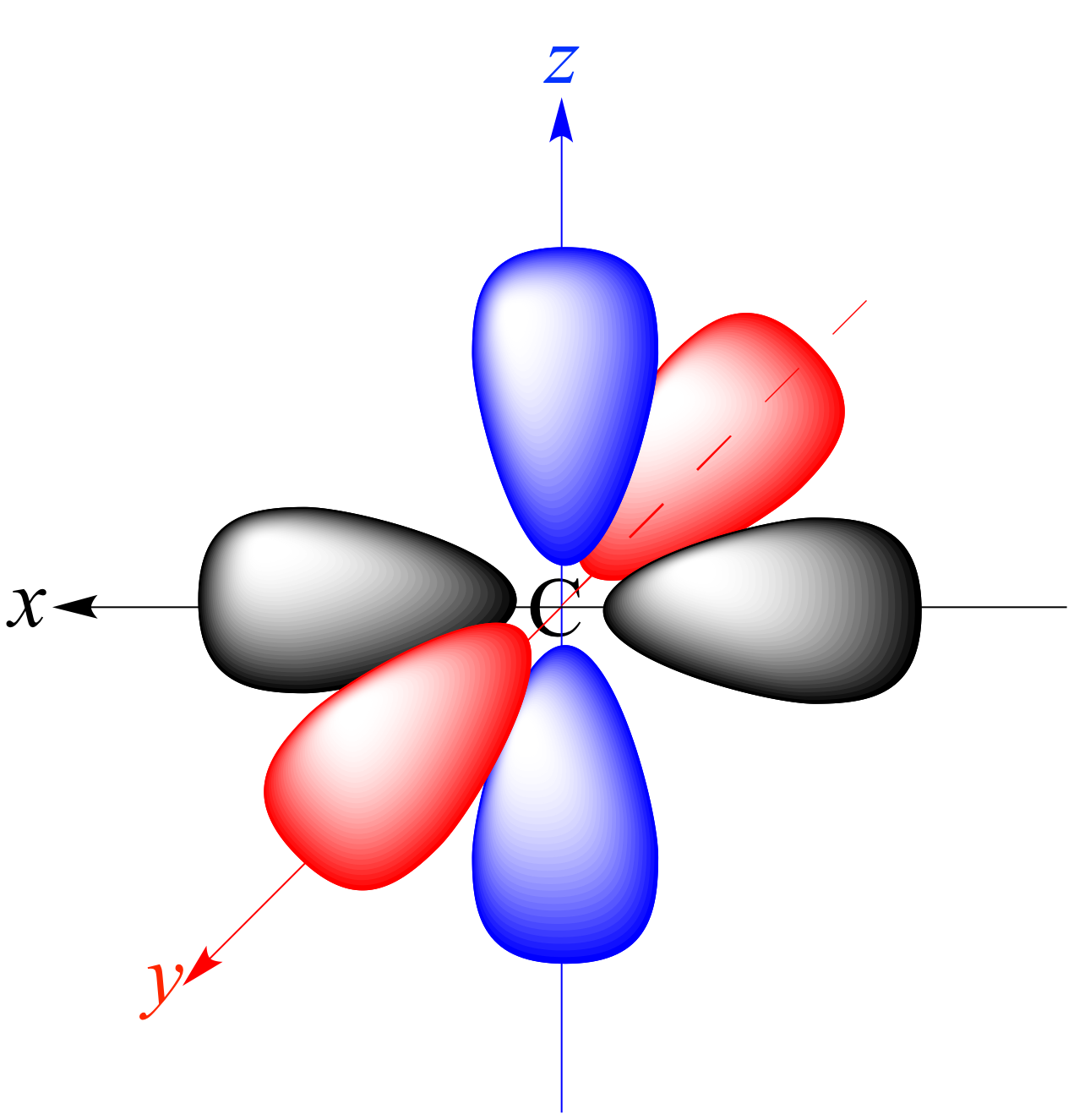
Figure 9.11. An sp 3 hybridized atomic orbital.Īccording to VSEPR theory, the four degenerate orbitals will arrange as far apart from each other as possible, giving a tetrahedral geometry with each orbital 109.5 o apart (Figure 9.12 “ A carbon atom’s four tetrahedral sp 3 hybridized orbitals”). This larger lobe is typically used for orbital overlap in covalent bonding. The sp 3 orbitals, being a combination of a spherical s orbital and propeller- (or peanut-) shaped p orbital, give an unsymmetrical propeller shape where one lobe of the orbital is larger (fatter) than the other (Figure 9.11 “ An sp 3 hybridized atomic orbital”). Hybridization of carbon to generate sp 3 orbitals. Note that in hybridization, the number of atomic orbitals hybridized is equal to the number of hybrid orbitals generated. When two half filled atomic orbitals belonging to two same or different atoms are brought near to each other. Moreover, the Valence Shell Electron Pair Repulsion (VSEPR) theory, says the molecular geometry of a molecule is trigonal planar. The 2 s and three 2 p orbitals are averaged mathematically through hybridization to produce four degenerate sp 3 hybrid orbitals (Figure 9.10 “ Hybridization of carbon to generate sp 3 orbitals”). The CH2O is a tetra atomic molecule where the bond angles for the hydrogen-carbon-hydrogen (H-C-H) and hydrogen-carbon-oxygen (H-C-O) are 116 and 122 and the structure is bent shaped. In 1931, Linus Pauling (Figure 9.9) proposed a mathematical mixing of atomic orbitals known as hybridization. Pi bonds result from the sideways overlap of p orbitals, placing electron density on opposite sides of the internuclear axis (Figure 9.7 “Pi bond diagram showing sideways overlapping of p orbitals”).įigure 9.9. A diagram showing the overlap of s orbitals of two hydrogen atoms to form H 2.įor molecules that contain double or triple bonds, one of these bonds is a sigma bond, and the remaining multiple bonds are a different type of bond known as a pi bond (π bond). The optimal distance between atoms, which maximizes the attractive forces and minimizes the repulsive forces, gives the H-H sigma bond a length of 74 pm. When carbon atoms make use of sp 2 hybrid orbitals for sigma bonding, the three bonds lie on the same plane. Repulsion forces between the two nuclei and between the two electrons are also present. The 1 s orbitals of the two hydrogens approach each other and overlap to form a bond that has cylindrical symmetry known as a sigma bond (σ bond). Let’s examine the simplest case of atomic overlap resulting in a covalent bond, the formation of H 2 from two hydrogen atoms (Figure 9.6 “ A diagram showing the overlap of s orbitals of two hydrogen atoms to form H 2“). Since these electrons are simultaneously attracted to both nuclei, the electron pair holds the two atoms together. 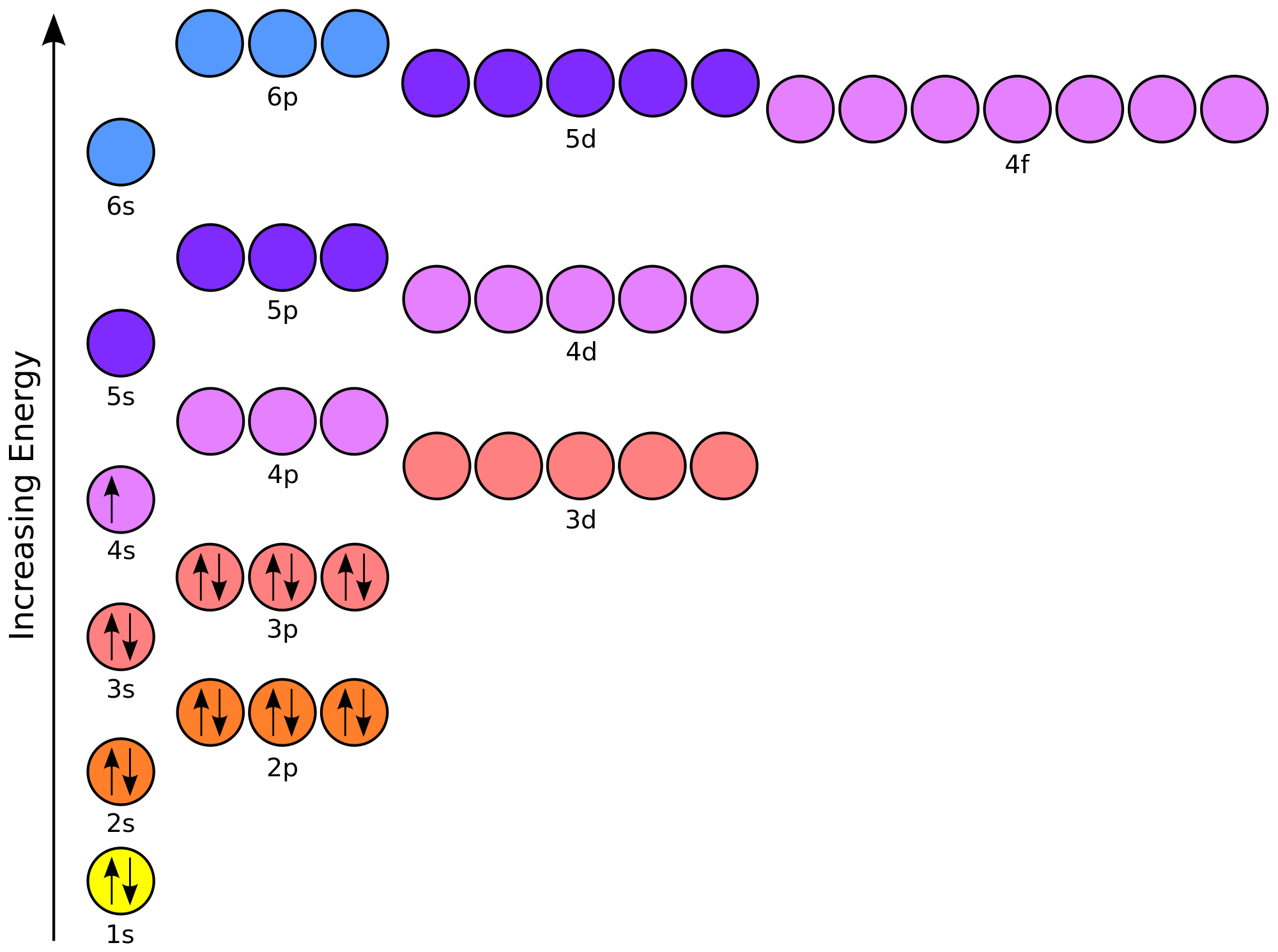
This creates an area of electron pair density between the two atoms. 
Depending on how many valence electrons each building. The valence bond theory states that atoms in a covalent bond share electron density through the overlapping of their valence atomic orbitals. The bonding through antibonding energies and band orbitals arising from s and from p atomic orbitals. Representations of s and p atomic orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
